- Blog
- Blog
- Gym leader raihan
- 2007 h3 hummer
- Keilwerth tenor sax kirk whalum
- Kairosoft games for free
- Slime rancher ps4
- Breakaway audio enhancer free
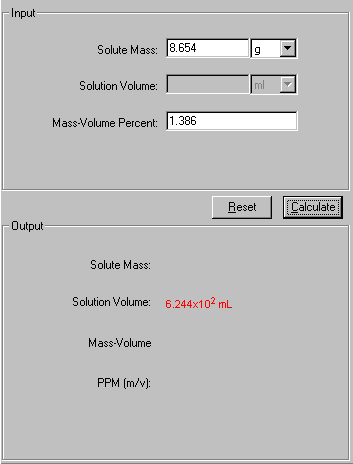
- How to calculate ppm by mass
- Pandigital scanner mac panscan05
- Maxmind db
- Dragon ball xenoverse mods ps3
- Raspberry pi youtube view bot
- Firewatch day 78
- Durango wild lands fiber
The concentration expressed in terms of the mass of the contaminants per cubic meter of the atmosphere is called the mass-volume concentration, its unit is milligrams per cubic meter or gram / cubic meter.įrom the formula: X-contaminant is expressed as the concentration value in milligrams per cubic meter C-concentration value of contaminants is expressed by ppm M-the molecular weight of contaminants. In the case of using ppm as the concentration of contaminants, this problem does not occur for it is volume ratio.Ĭoncentration unit ppm and mg / m3 conversion: The use of mass concentration unit (mg / m3) as the air pollutant representation method, we can easily calculate the true amount of pollutants.īut the quality concentration has some relationship with gas temperature, pressure and environmental conditions, and its value will be different with temperature, air pressure and other environmental conditions variation the actual measurement needs simultaneously measure the gas temperature and atmospheric pressure.
#How to calculate ppm by mass how to#
What is the relationship between these two gas concentration units in mg / m3 and ppm? How to convert them?

Our national standards are also expressed in mass concentration units (eg mg / m3). Mass concentration representation: the mass per ton of air contained in the air mg / m3 volume concentration represents: one million volumes of air contained in the volume of pollutants, that is, concentration results of most of the gas detection equipment is volume concentration (ppm).Īccording to our regulations, especially Environmental Protection Dept., gas concentration is required to be expressed in units of mass concentration (eg mg / m3). There are two ways to represent pollutant concentrations in ambient air (air): That is, ppm = 35 (% LEL) * 2 (vol%) * 100 = 7000 ppm methaneĬonversion relationship of gas detection concentration unit: ppm and milligrams / cubic meter

I.e.: 35% LEL of methane, its LEL is 2vol% Ppm unit can be converted to LEL as follows: For example, lower limit of explosion of methane is 5% VOL, so 10% LEL methane gas has the following correspondence: 10% LEL = 5000ppm = 0.5% VOL The relationship between the three units: In normal cases, ppm is used for accurate measurement LEL is used for explosion test magnitude of VOL is the largest of the three units. I.e.: 5% VOL means the specific gas occupies 5% volume of air. I.e.: 25LEL% means 25% of lower limit of explosionĥ0LEL% means 50% of the lower limit of explosionģ.VOL: gas volume percentage, it is a physical unit. LEL% Percentage of lower limit of explosion, that is, the lower limit of the explosion is divided into one hundred parts, one unit is 1LEL%. (UEL: upper limit concentration of gas explosion.) LEL: the minimum volume percentage concentration of combustible gas in air that can detonate, that is, the lower limit of gas explosion concentration. I.e.: 5ppm carbon monoxide means that the air contains 5 per million of carbon monoxide.Ģ. ppm: gas volume percentage per million, it is a dimensionless unit.
The meaning and conversion of ppm, LEL and VOLġ. If you cannot understand from above, please see the following specific explanation: The relationship between LEL and VOL is defined according to the standard, for example, the lower limit of methane concentration in China is defined as 5Vol%, while the lower limit of explosion in Germany is set to be more conservative and rigorous, that is 4.4Vol%. It is worth noticing that this unit is not a natural unit, but the lower limit of the explosion is divided into one hundred parts, one part for 1% LEL.

% LEL means the lower limit of explosion, that is, in which concentration will the combustible gas explode when it meets fire. PPM unit can be converted to LEL as follows:įor example: 35% LEL of methane, its LEL is 2 vol%, that is, ppm=35(%LEL)*2(vol%)*100=7000ppm methaneģ00ppm of propane, its LEL is 1.2vol5, that is, %LEL=300ppm/(1.2 vol%*100)=2.5% LEL propane
